Category: teaching
New Masterclass: “Papers and Books”
 “Navigating numbers” is a series of Masterclass initiated by a team of Charité researchers who think that our students should be able to get more familiar how numbers shape the field of medicine, i.e. both medical practice and medical research. And I get to organize the next in line.
“Navigating numbers” is a series of Masterclass initiated by a team of Charité researchers who think that our students should be able to get more familiar how numbers shape the field of medicine, i.e. both medical practice and medical research. And I get to organize the next in line.
I am very excited to organise the next Masterclass together with J.O. a bright researcher with a focus on health economics. As the full title of the masterclass is “Papers and Books – series 1 – intended effect of treatments”, some health economics knowledge is a must in this journal club style series of meetings.
But what will we exactly do? This Masterclass will focus on reading some papers as well as a book (very surprising), all with a focus on study design and how to do proper research into “intended effect of treatment” . I borrowed this term from one of my former epidemiology teachers, Jan Vandenbroucke, as it helps to denote only a part of the field of medical research with its own idiosyncrasies, yet not limited by study design.
The Masterclass runs for 8 meetings only, and as such not nearly enough to have the students understand all in and outs of proper study design. But that is also not the goal: we want to show the participants how one should go about when the ultimate question is medicine is asked: “should we treat or not?”
If you want to participate, please check out our flyer
Starting a research group: some thoughts for a new paper

It has been 18 months since I started in Berlin to start at the CSB to take over the lead of the clinical epidemiology research group. Recently, the ISTH early career taskforce have contacted me whether I would be willing to write something about my experiences over the last 18 months as a rookie group leader. The idea is that these experiences, combined with a couple of other papers on similar useful topics for early career researchers, will be published in JTH.
I was a bit reluctant at first, as I believe that how people handle new situations that one encounters as a new group leader is quite dependent on personality and the individual circumstances. But then again, the new situations that i encountered might be more generalizable to other people. So I decided to go ahead and focus more on the description of the new situations I found myself in while trying to keep the personal experiences limited and only for illustrations only.
While writing, I have discerned that there are basically 4 new things about my new situations that I would have loved to realise a bit earlier.
- A new research group is never without context; get to know the academic landscape of your research group as this is where you find people for new collaboration etc
- You either start a new research group from scratch, or your inherit a research group; be aware that both have very different consequences and require different approaches.
- Try to find training and mentoring to help you cope with your new roles that group leaders have; it is not only the role of group leader that you need to get adjusted to. HR manager, accountant, mentor, researcher, project initiator, project manager, consultant are just a couple of roles that I also need to fulfill on a regular basis.
- New projects; it is tempting to put all your power, attention time and money behind a project. but sometimes new projects fail. Perhaps start a couple of small side projects as a contingency?
As said, the stuff I describe in the paper might be very specific for my situation and as such not likely to be applicable for everyone. Nonetheless, I hope that reading the paper might help other young researchers to help them prepare for the transition from post-doc to group leader. I will report back when the paper is finished and available online.
Statins and risk of poststroke hemorrhagic complications
 Easter brought another publication, this time with the title
Easter brought another publication, this time with the title
“Statins and risk of poststroke hemorrhagic complications”
I am very pleased with this paper as it demonstrates two important aspects of my job. First, I was able to share my thought on comparing current users vs never users. As has been argued before (e.g. by the group of Hérnan) and also articulated in a letter to the editor I wrote with colleagues from Leiden, such a comparison brings forth an inherent survival bias: you are comparing never users (i.e. those without indication) vs current users (those who have the indication, can handle the side-effects of the medication, and stay alive long enough to be enrolled into the study as users). This matter is of course only relevant if you want to test the effect of statins, not if you are interested in the mere predictive value of being a statin user.
The second thing about this paper is the way we were able to use data from the VISTA collaboration, which is a large amount of data pooled from previous stroke studies (RCT and observational). I believe such ways of sharing data brings forward science. Should all data be shared online for all to use? I do am not sure of that, but the easy access model of the VISTA collaboration (which includes data maintenance and harmonization etc) is certainly appealing.
The paper can be found here, and on my mendeley profile.
– update 1.5.2016: this paper was topic of a comment in the @greenjournal. See also their website
update 19.5.2016: this project also led to first author JS to be awarded with the young researcher award of the ESOC2016.
We congratulate our @Jan_FriSch with his young investigator award #ESOC2016 pic.twitter.com/eCqXiw0Ub1
— CSB (@BerlinStroke) 10 May 2016
Where Have All the Rodents Gone? The Effects of Attrition in Experimental Research on Cancer and Stroke
We published a new article just in PLOS Biology today, with the title:
“Where Have All the Rodents Gone? The Effects of Attrition in Experimental Research on Cancer and Stroke”
This is a wonderful collaboration between three fields: stats, epi and lab researchers. Combined we took a look at what is called attrition in the preclinical labs, that is the loss of data in animal experiments. This could be because the animal died before the needed data could be obtained, or just because a measurement failed. This loss of data can be translated to the concept of loss to follow-up in epidemiological cohort studies, and from this field we know that this could lead to substantial loss of statistical power and perhaps even bias.
But it was unknown to what extent this also was a problem in preclinical research, so we did two things. We looked at how often papers indicated there was attrition (with an alarming number of papers that did not provide the data for us to establish whether there was attrition), and we did some simulation what happens if there is attrition in various scenarios. The results paint a clear picture: the loss of power but also the bias is substantial. The degree of these is of course dependent on the scenario of attrition, but the message of the paper is clear: we should be aware of the problems that come with attrition and that reporting on attrition is the first step in minimising this problem.
A nice thing about this paper is that coincides with the start of a new research section in the PLOS galaxy, being “meta-research”, a collection of papers that all focus on how science works, behaves, and can or even should be improved. I can only welcome this, as more projects on this topic are in our pipeline!
The article can be found on pubmed and my mendeley profile.
Update 6.1.16: WOW what a media attention for this one. Interviews with outlets from UK, US, Germany, Switzerland, Argentina, France, Australia etc, German Radio, the dutch Volkskrant, and a video on focus.de. More via the corresponding altmetrics page . Also interesting is the post by UD, the lead in this project and chief of the CSB, on his own blog “To infinity, and beyond!”
New article published – Ankle-Brachial Index and Recurrent Stroke Risk: Meta-Analysis

Another publication, this time on the role of the ABI as a predictor for stroke recurrence. This is a meta analysis, which combines data from 11 studies allowing us to see that ABI was moderately associated with recurrent stroke (RR1.7) and vascular events (RR 2.2). Not that much, but it might be just enough to increase some of the risk prediction models available for stroke patients when ABI is incorperated.
This work, the product of the great work of some of the bright students that work at the CSB (JBH and COL), is a good start in our search for a good stroke recurrence risk prediction model. Thiswill be a major topic in our future research in the PROSCIS study which is led by TGL. I am looking forward to the results of that study, as better prediction models are needed in the clinic especially true as more precise data and diagnosis might lead to better subgroup specific risk prediction and treatment.
The article can be found on pubmed and my mendeley profile and should be cited as
Hong J Bin, Leonards CO, Endres M, Siegerink B, Liman TG. Ankle-Brachial Index and Recurrent Stroke Risk. Stroke 2015; : STROKEAHA.115.011321.
New article published – Conducting your own research: a revised recipe for a clinical research training project
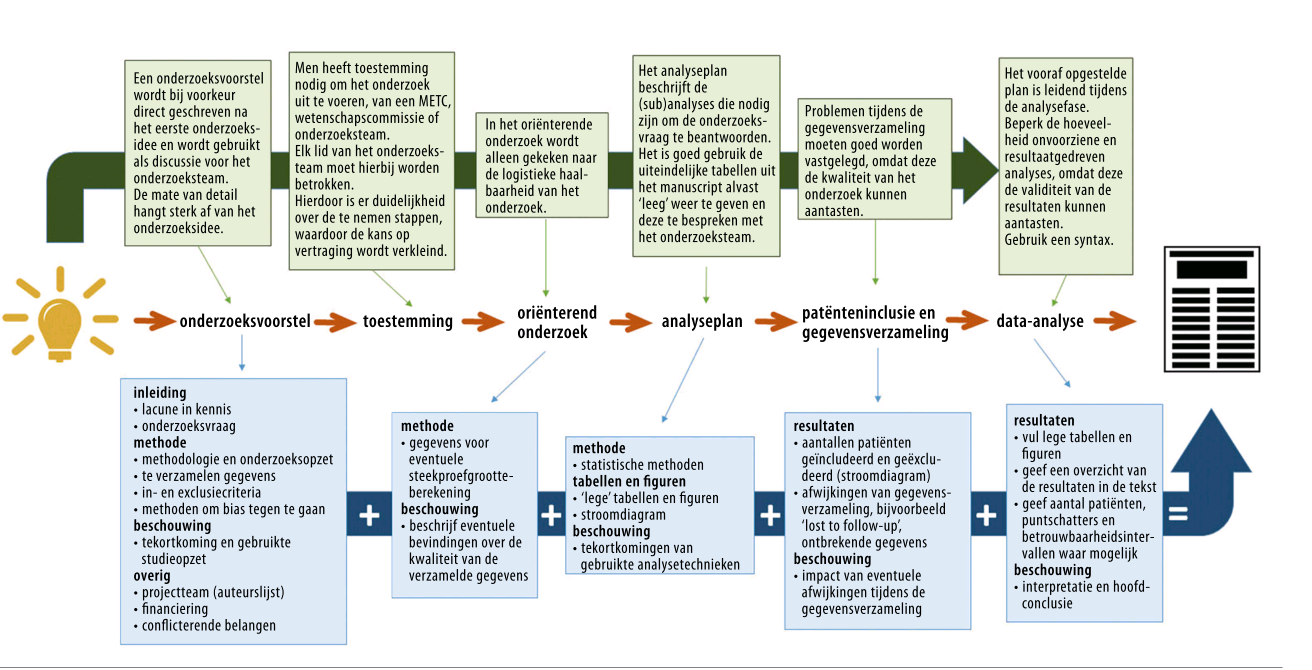
A quick update on a new article that was published on friday in the NTVG. This article with the title
“Conducting your own research: a revised recipe for a clinical research training project”
– gives a couple of suggestions for young clinicians/researchers on how they should organise their epidemiological research projects. This paper was written to commemorate the retirement of prof JvdB, who wrote the original article back in 1989. I am quite grew quite fond of this article, as it combines insights from 25 years back as well as quite recent insights (e.g. STROBE and cie Schuyt and resulted in a article that will help young research to rethink how they plan and execute their own research project.
There are 5 key suggestions that form the backbone of this article i.e. limit the research question, conduct a pilot study, write the article before you collect the data, streamline the research process and be accountable. As the article is in Dutch only at this moment, I will work on an English version. First drafts of this ms, each discussing each of the 5 recommendations might appear on this website. And how about a German version?
Anyway, it has to be mentioned that if it not was for JvdB, this article would have never come to light. Not only because he wrote the original, but mostly because he is one of the most inspiring teachers of epidemiology.
Journal Club 2015
A new semester, time for new cool stuff. I am starting a Epi Journal Club for medical students. Here is the recruitment text.
Do you want to learn how to read medical literature?
What is a Kaplan Meier Curve? What is the difference between a case-control study and a case-cohort study? How do you get rid of confounding? What is confounding anyway! Or just in short, what distinguishes a good paper from a bad paper? Do you want to know? Here is a start:
Bob Siegerink is a clinical epidemiologist from the Centrum für Schlaganfallforschung Berlin (CSB) and he is starting a journal club for medical students (5th semester and up) where participants will be discussing both good and bad papers from both ancient and recent history. Although there is no previous experience needed, students have to be highly motivated as this is a fun, but hardcore, crash course in clinical epidemiology. All discussion will be held in English.
I have no clue on how many student will react: the deadline is soon, but this text will end up in the inbox of a lot of students. And as the level of science education in their curricula is limited compared to the Dutch curricula, they might not be triggered by this topic. Or they will… Who knows? I will use some of my previous teaching material, but I guess I need to adapt the content based on the number of participants and their level or knowledge, so everything is possible. I will keep you posted!
– update 18-4-2015: 11 participants… not bad for a first time! we start in 10 days. For the programme, click here
New publication “Graphical presentation of confounding in directed acyclic graphs”

A new publication became available, again an ‘educational’. However, this time the topic is new. It is about the application of directed acyclic graphs, a technique widely used in different areas of science. Ranging from computer science, mathematics, psychology, economics and epidemiology, this specific type of graphs has shown to be useful to describe the underlying causal structure of mechanisms of interest. This comes in very handy, since it can help to determine the sources of confounding for a specific epidemiological research question.
But, isn’t that what epidemiologist do all the time? What is new about these graphs, except for the fancy concepts as colliders, edges, and backdoor paths? Well, the idea behind DAGs are not new, there have been diagrams in epidemiology since years, but each epidemiologist has his own specific ways to draw the different relationship between various variables factors. Did you ever got stuck in a discussion about if something is a confounder or not? If you don’t get it resolved by talking, you might want to draw out the your point of view in a diagram, only to see that your colleagues is used to a different way of drawing epidemiological diagrams. DAGs resolve this. There is a clear set on rules that each DAG should comply with and if they do, they provides a clear overview of the sources of confounding and identify the minimal set of variables to account for all confounding present.
So that’s it… DAGs are a nifty method to talk the same idiom while discussing the causal questions you want to resolve. The only thing that you and your colleague now can fight over is the validity of the assumptions made by the DAG you just drew. And that is called good science!
The paper, with first author MMS, appeared in the methodology series of the journal Nephrology Dialysis and Transplantation, can be found here in pdf, and also on my mendeley account.
preconference workshop ‘crash course peer review’ cancelled
I worked together with some partners on a new workshop for young epidemiologist. The title says it all: WEON preconference workshop ‘crash course peer review’.
Unfortunately, we had to cancel the workshop because the number of participants was to low to justify the effort of not only myself, but especially all the other teachers. I think it is a pity that we had to cancel, but by cancelling we still have a fresh start whenever we want to try again in a different format.
Whilst preparing this workshop I noticed that peer review, or a better term would be refereeing, is not popular. It is seen as a task that task up to much time, with too much political consequences and little reward etc. New initiatives like Pubmed commons and other post publication peer review systems are regarded by some as answers to some of these problems. But what is the future of refereeing, when young epidemiologist are not intrinsically motivated to contribute time and effort to the publication process? Only time will tell.
For those who are still interested in this crash course, please contact me via email.
Bad Pharma 2013 – a great succes

300 students reading one book, summarising it contents and discussing three propositions in order to grasp the essence of the book. This might sound a bit boring perhaps…But what if it Goldacres book Bad Pharma, you only have to read 30 pages and still get the content of the complete book, and get to Skype with the author? Great fun!
And we weren’t the only who had fun: Ben Goldacre addressed 300 students, all with critical questions, and apparently he enjoyed the whole debate:
We also invited AV, a spokesperson of NEFARMA an organisation who represents most pharmaceutical companies in the Netherlands. With him we discussed whether the AllTrials campaign was a waste of time and money. Also, BB a reporter from the Mare, who has knowledge on conflict of interest of professors joined in on the discussion whether professors should declare their COI before each lecture. All in all, it was a great day in which some of the students were shocked by the message of Ben Goldacre, whereas other students were not impressed and doubted whether there view is too one-sided… The debate still continues!
Bad Pharma Symposium on the front page of Mare
 This week, the Mare decided to run a story on Bad Pharma book by Ben Goldacre and our related symposium. The author, BB did an outstanding job in describing the argument Goldacre brings forward in his book. As you might know, we are organising a symposium for our 300 students that are following our course “academic and scientific training”, because I believe that doctors should learn about their field that they will graduate in once they have graduated. A quote from me in the Mare (in Dutch)
This week, the Mare decided to run a story on Bad Pharma book by Ben Goldacre and our related symposium. The author, BB did an outstanding job in describing the argument Goldacre brings forward in his book. As you might know, we are organising a symposium for our 300 students that are following our course “academic and scientific training”, because I believe that doctors should learn about their field that they will graduate in once they have graduated. A quote from me in the Mare (in Dutch)
Dokter zijn gaat verder dan alleen het behandelen van één patiënt. Onze beroepsgroep heeft een bijzondere positie in de samenleving; mensen leggen letterlijk hun leven in je handen. Naast je arts-patiëntrelatie heb je ook te maken met de wetenschap, beleidsmakers en de farmaceutische industrie. Aankomende artsen moeten daarvan bewust worden en goed op de hoogte zijn van de ontwikkelingen in hun veld. Dit boek sloeg de spijker op zijn kop.
one little error slipped in… I am not a doctor and therefore it is officially not ‘onze beroepsgroep’ but i think people will grasp the point that I try to make. The complete article can be read here.
Academic and scientific training about to start
After 7 months of preparation the new and improved version of the second year course ‘Academic and Scientific Training’ is about to start. Tomorrow, 300 students will come together for two weeks to learn the how and why of clinical epidemiology. We emphasize on testing new treatments in RCT, and how doctors should critically appraise the articles that describe these studies.
Together with AvHV, JS and others we were able to secure funding to buy 400 copies of the book “Bad Pharma” (click for more background) In order to organise a ‘Bad Pharma Symposium’. In this symposium, all students will read the whole book, do some research on three topics which will be debated in the final debate. This will take a lot of commitment of all students, but what to think of the 40+ Teaching Assistants that we need for this course!
All in all, it’s time to start!
ERA-EDTA CME course – “interpretation and presentation of study results”

During “conference season” I visited several conferences: ISTH, eurostroke, WEON, NVTH, UK-CIM and the ERA-EDTA. During all conferences I got the opportunity to present my own research, except for during the ERA-EDTA. For this conference I was asked to teach in a CME course on how to perform and interpret a clinical research project. The program:
– Setting up your study: study questions and study designs
Vianda Stel, Amsterdam, the Netherlands
– Threats to validity of study findings: bias and confounding
Kitty Jager, Amsterdam, the Netherlands
– Prognosis vs aetiology
Friedo Dekker, Leiden, the Netherlands
– Interpretation and presentation of study results
Bob Siegerink, Leiden, the Netherlands
Since this is a conference on kidney diseases, the examples that I use are from that field. Although not necessarily my field, I believe that the talk can be of interest for anybody who is at the start of their research career. Please click the picture below to see the talk (slides + audio)
Bad Pharma Symposium
 I just got word that we got funding from an external to buy 400 copies of Bad Pharma to use as instruction material in our redesigned course on academic and scientific education. The book touches upon the role of the pharmaceutical industry in the design, execution, presentation and dissemination of results of clinical trials. The author, Ben Goldacre, identifies several problems and brings forth several solutions. But are these all correct?
I just got word that we got funding from an external to buy 400 copies of Bad Pharma to use as instruction material in our redesigned course on academic and scientific education. The book touches upon the role of the pharmaceutical industry in the design, execution, presentation and dissemination of results of clinical trials. The author, Ben Goldacre, identifies several problems and brings forth several solutions. But are these all correct?
We will use this basic question as the basis for a complete symposium on this topic: are the problems identified by Goldacre the real problems, or just cherry picked examples from the past? And are the solutions that he provides real solution that could work? And can we think of any other way to improve the care of our patients in the future?
I had this idea a long time ago when i first read this book. It touches upon a matter that is discussed in the old version of this course for a couple of years. Even more so: during this course students are asked to grade the quality of trials and the subsequent reference to this publication. The results show that the quality is often low, even in high impact journals, and that the references do not always justify the claims made in medical advertisements. These results have been published in several publications and are even cited in the book.
It was quite a hassle to get a ‘normal’ book into the curriculum of our medical center. Most people liked the idea of reading a book and organising a symposium, but a lot of people told me that it was impossible to do so. A “normal” book could not be placed on the mandatory reading list, and just buying electronic or hard copy versions is just way to expensive. luckily, with help from the Walaeus Library of the LUMC and prof FMH we were able to obtain external funding. And no, its not funded by ‘big pharma’, but a small fund that subsidises small but nice projects that make the world a bit smarter.
New course started today: Scientific education
Today, the course scientific education started. This course, a regular 2nd year course in the LUMC curriculum, runs for three weeks and is organised by AvHV and myself. This is the last year this course will be given in this form, and we are working hard to modify the course to make it fit in the new curriculum. This means that this year we have a opportunity to test some of my our ideas. One of these things is a non compulsory journalclub, in which we’re going to read one article with all students at the same time. I’m curious how may students really would like to train their scientific skills, outside of the curriculum.
Also, we are preparing a new full day interactive assignment and sympium, which is centered around the book Bad Pharma of author Ben Goldacre. I hope to fine some students that are wiling to help me in this development of this part of the new course. Exciting stuff!
– edit 30/5 : the course just finished and 320 students took their exam friday 24th. Although i do like students, I hope I won’t have to see them again for that will mean that they failed the course.
Hora Est – thesis is topic of a Cicero article
 The Cicero, the monthly magazine of the LUMC, wrote an article on my thesis. During the interview, I was able to bring fort the nuances of the work in the thesis. That is, my thesis does not fully provide all the answers in details for allr esearch questions but it does provide a simple overview: an increased clotting potential is a risk factor for ischaemic stroke but not for moycardial infarction. Unfortunately, space limitations dictated that some remarks had to be left out. Fortunately, the last remarks is about the relevance of teaching experience as a PhD: a great link to one of my propositions.
The Cicero, the monthly magazine of the LUMC, wrote an article on my thesis. During the interview, I was able to bring fort the nuances of the work in the thesis. That is, my thesis does not fully provide all the answers in details for allr esearch questions but it does provide a simple overview: an increased clotting potential is a risk factor for ischaemic stroke but not for moycardial infarction. Unfortunately, space limitations dictated that some remarks had to be left out. Fortunately, the last remarks is about the relevance of teaching experience as a PhD: a great link to one of my propositions.
The picture above was placed in the Cicero to accompany the interview. I like it very much because it has a certain epidemiological feel to it: a group (a cohort?) of women all different, but with certain patterns.
The text of the interview can be downloaded from the media page, directly directly clicking here (pdf). The text can also be read online if you… Continue reading “Hora Est – thesis is topic of a Cicero article”
New article accepted for publication in NTVG

A new article has been accepted in the Nederlands Tijdschrift voor Geneeskunde. The article with the title “patient crossover studies” or “case-crossover studies” is an educational in the Methodology series of the Journal. REJR is the first author of this article and she did a great job on explaining the similarities and differences between this observational study design and the experimental version of this within person comparison. These crossover trials have been discussed by TNB en JGvdB in a previous article in the same series on which i wrote earlier.
Ben Goldacres ‘Bad Pharma’ and research from the LUMC

Ben Goldacre, known from the bestseller Bad Science (book and blog) has a new book, Bad Pharma. Goldacre is always fun to read: science, both the method as the social phenomenon, explained for non-scientist while still interesting for scientist. The same goes for his new title Bad Pharma, where he explains what is right and wrong in the field of clinical trials needed to determine what treatment is best given. Before I am going to review the complete book, perhaps this TED talk will explain it all:
Basically, his point is that for good answers to questions on what treatment is best to save lives, it is pivotal that all the results of all trials are published. This sounds a bit old, since there are databases in which trials should be registered. However, only registering the existence of a trial is not enough: all data should become known to the public. This sounds familiar: this standpoint is off course the same standpoint of the AllTrials.net petition, which is initiated by a.o. Ben Goldacre. For more on AllTrials.net, please see a previous post.
While reading the book of Goldacre it started reading about reasearch done in the Netherlands, where 250 students were looking into the adverts for medication: they checked their quality (was the science OK?) and correct use (does it support the claim?) of the trials in major journals and found that half was of good quality and only half supported the claim. And the nice thing about this research? It was executed at our department as part of one of our undergraduate courses! All students scored trials and a couple of students were also engaged in the analyses/writing/submission process. The paper from this research, cited by Goldacre, is available from the website of the Netherlands Journal of Medicine. (pdf, open access) An earlier paper with the same concept but focussed on rheumatoid arthritis medication is also published, also open acces. (pdf)
Masterclass “Noordwijk” covered in the LUMC magazine Cicero

The LUMC magazine “Cicero” covered our Masterclass in Noordwijk. Its a nice description (in Dutch) of two weekend of undergrad-die-hard-epidemiology. One of the students is also interviewed and she concludes:
“Het lukt de docenten om de studenten de hele tijd
te blijven boeien, gedurende twee weekenden van donderdagavond tot zaterdagmiddag. Ik was bang dat ik dat niet zou volhouden. Maar het ging, en het bleef nog leuk ook.”
The text of the article can be found below and here in pdf (cicero 29 jan 2013). More articles etc can be found on the media page.
Continue reading “Masterclass “Noordwijk” covered in the LUMC magazine Cicero”
Poster on ERA EDTA courses
A new poster has been designed to promote the ERA-EDTA epidemiology courses. I think the poster looks great, and it will certainly help to recruit new participants at the meeting in Istanbul.

ERA-EDTA Istanbul
 The 50th edition of the ERA EDTA congress will he be held in Istanbul from the 18th until the 21st in may. Since I mainly work i the field of thrombosis, I have never been to this congress, but I probably will visit it next year: The epidemiological team from the AMC is invite to organise an educational session on clinical epidemiology and they asked me to join their team. The content of this methodological session will resemble the smaller meeting we had last year in Thessaloniki, Greece.
The 50th edition of the ERA EDTA congress will he be held in Istanbul from the 18th until the 21st in may. Since I mainly work i the field of thrombosis, I have never been to this congress, but I probably will visit it next year: The epidemiological team from the AMC is invite to organise an educational session on clinical epidemiology and they asked me to join their team. The content of this methodological session will resemble the smaller meeting we had last year in Thessaloniki, Greece.
Also, I will also be scoring some abstracts for this meeting: I got an email telling me, or warning me perhaps, to expect up to 80 abstract for me to score. In just a couple of days. Let’s hope that they have clear guidelines to help, otherwise it will be tough nut to crack. Nonetheless, both activities give me some insight in the field of nephrology and will help me to further train my epidemiological teaching skills.
Opzet en interpretatie van mensgebonden onderzoek – slides online
Tomorrow I will teach at the graduate course ‘Design and analysis of clinical research’. My part is to introduce the concept of confounding which i demonstrate through the general idea of ‘confusing of effects’. Perhaps a bit ‘oldskool’, but it works as a nice introduction to the concept without a direct confrontation with DAGs etc, especially since it helps to think in ways to prevent / solve this problem in data analyses. What ‘arrow’ in the classic confounding triangle can be removed?
I also go into the concept of ceteris paribus, which is further explored through examples of IV analyses. These examples can be historical (Boylston and inoculation) or recent (mendelian randomisation on CRP and CVD disease).
Slides are present in the presentations section of this website.


